1. Introduction
The lithospheric mantle is rarely preserved in a pristine, anhydrous state due to pervasive hydration processes. Recent studies have increasingly recognized hydration as a key mechanism altering mantle rocks, primarily through melt or fluid metasomatism, which introduces water and drives significant modifications in metal isotope compositions (e.g., Cui et al., 2025; Demouchy et al., 2015; J. Liu et al., 2017; Nishi, 2015; Schmädicke et al., 2018; B.-X. Su et al., 2018, 2021, 2025). However, stable metal isotope distributions, particularly Fe and Zn, exhibit striking differences. In continental mantle peridotite xenoliths, Fe isotopes follow a systematic equilibrium fractionation sequence (δ56Fe: spinel > clinopyroxene > orthopyroxene > olivine), consistent with bonding strength controls (Beard & Johnson, 2004; Huang et al., 2011; Macris et al., 2015; Roskosz et al., 2015; Schauble et al., 2001; Williams et al., 2005; Zhao et al., 2012, 2015, 2017). In contrast, ophiolitic mantle peridotites display a reversed Fe isotope sequence, with silicates enriched in heavier isotopes relative to coexisting spinel (Chen et al., 2015; B.-X. Su et al., 2021; Xiao et al., 2016; Zhang et al., 2017). This anomaly extends to Zn and Cr isotopes, which similarly deviate from theoretical predictions (Chen et al., 2019; Fang et al., 2022; Shen et al., 2018; Z.-Z. Wang et al., 2017; Yang et al., 2021).
These reversed isotope fractionations, consistently observed across ophiolites, challenge conventional models of mantle processes and suggest a distinctive characteristic of the oceanic lithospheric mantle. Hydrous fluid-mediated element exchange, potentially linked to chromitite formation or subduction-related fluids, has been proposed as a key driver (Gose & Schmädicke, 2021; B. X. Su et al., 2021; B.-X. Su et al., 2020, 2021, 2023, 2026; in press; Xiao et al., 2023). Yet, the absence of significant chromite deposits in mid-ocean ridges and some ophiolites, coupled with variable fluid sources, indicates that hydration is a complex, multi-faceted process (Cui et al., 2025; B.-X. Su et al., 2023, 2025, 2026). The scale and impact of hydration have historically been underestimated, obscuring its role in reshaping mantle geochemistry. The mechanisms governing inter-mineral isotope fractionation and their connection to pervasive hydration remain unsolved, limiting our understanding of oceanic lithospheric mantle evolution.
This study presents high-precision Fe and Zn isotope data from coexisting minerals (spinel, orthopyroxene, clinopyroxene, and olivine) in mantle rocks from an ophiolite in Tibetan Plateau. By integrating these data with global ophiolite records, we elucidate the controls on inter-mineral isotope fractionation, revealing hydrous fluid-mediated element exchange as a widespread process in the oceanic lithospheric mantle. Our findings highlight the transformative role of hydration, demonstrating its profound impact on mantle geochemistry and necessitating a revised framework for interpreting oceanic mantle processes.
2. Purang ophiolite and samples
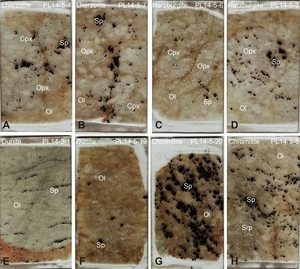
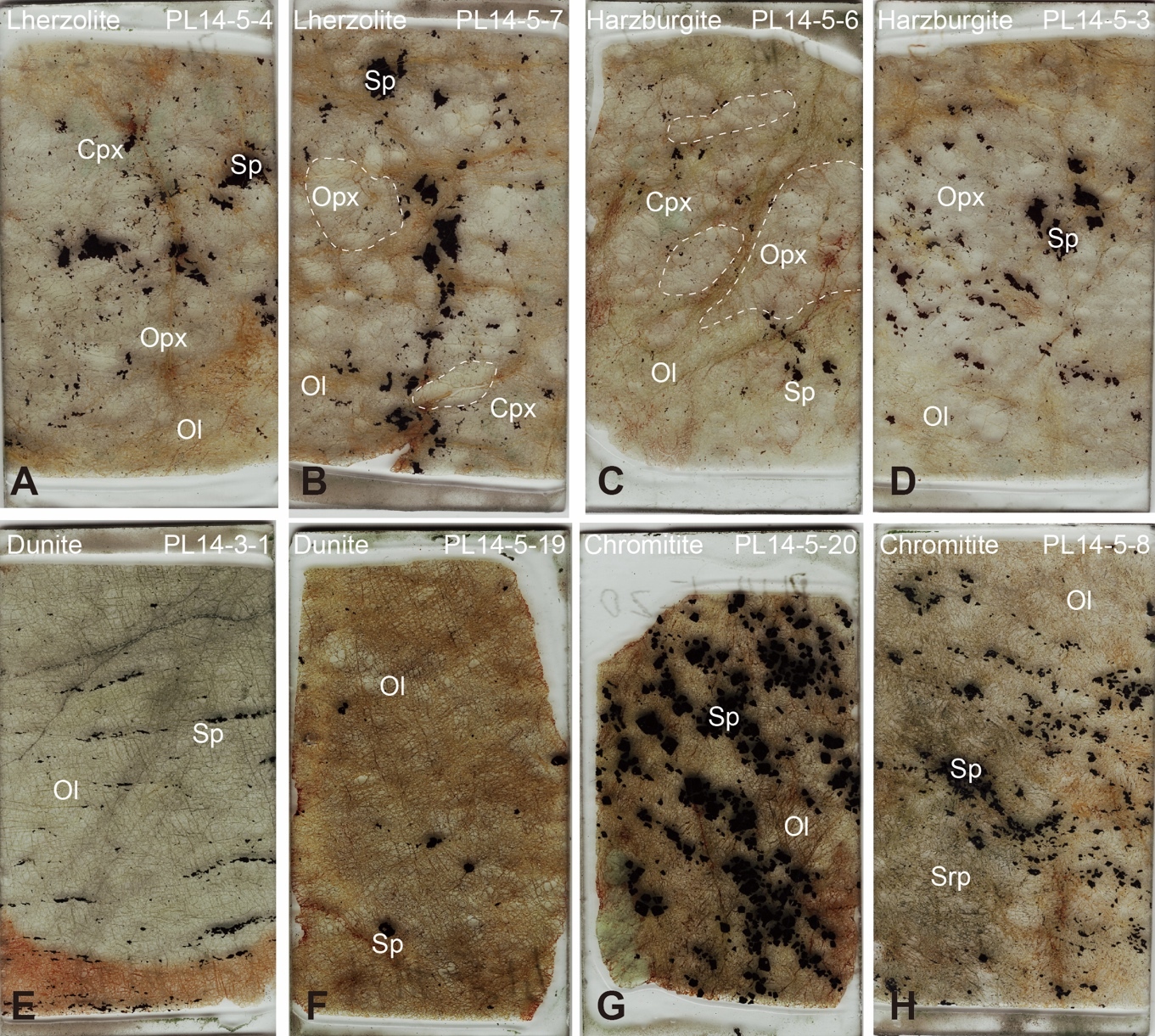
The Purang ophiolite is located in the Yarlung-Zangbo Suture zone in the Tibetan Plateau (fig. 1A, B). It exposes a well-preserved mantle sequence dominated by harzburgites with subordinate lherzolites and dunites (fig. 1C; C.-Z. Liu et al., 2010; F. Liu et al., 2015). Our samples, largely unaltered (figs. 2, S1) have been previously characterized for petrography and elemental geochemistry (B. X. Su et al., 2019; B.-X. Su et al., 2015) and are briefly summarized here. The lherzolites and harzburgites have similar mineral assemblages with differences in modal contents of clinopyroxene, and their orthopyroxene grains are highly variable in size from <1 mm to >2.5 cm (fig. 2A–D). The dunites and chromitites are commonly associated with each other and consist exclusively of olivine and spinel, with minor amounts of serpentine in a few samples (fig. 2E–H). Spinel, orthopyroxene, clinopyroxene, and olivine were separated from 4 lherzolites, 7 harzburgites, 6 dunites, and 5 chromitites from the Purang ophiolite for Fe and Zn isotope analyses. The major oxide mineral chemistry of the Purang lherzolites and harzburgites is very similar (B. X. Su et al., 2019; B.-X. Su et al., 2015). Olivine in the lherzolites and harzburgites has forsterite (Fo) contents of 90.5–92 and shows no apparent correlation with NiO contents, whereas olivine in the dunites and chromitites has a slightly larger variation in Fo (90–93) and exhibits a positive correlation between Fo and NiO. All spinel grains in the studied samples are high-Al varieties with Cr# (100×Cr/(Cr+Al)) values of 15.8 to 50.1. Orthopyroxenes generally have variable CaO and Al2O3 contents and Cr#s and show weak or no correlation with Mg# values (100×Mg/(Mg+Fe); 89.7 to 91.8). Clinopyroxene compositions in the Purang samples are highly variable, such as Mg# = 91.1 to 94.4 (B. X. Su et al., 2019).
3. Analytical methods
Fe-Zn isotopic analyses were conducted at Metallogenic Elements and Isotopes Laboratory in the Institute of Geology and Geophysics, Chinese Academy of Sciences, following the protocol described in J. Wang et al. (2022, 2023). 6–200 mg of minerals were weighed. The spinel was preprocessed using a M6 Microwave Oven (PreeKem, China) with mixture of concentrated 4.5 mL HCl, 1.5 mL HNO3, 3 mL HF and 1 mL HClO4. And then followed the general routine like other minerals concentrated HNO3 and HF acids in 7 mL PTFE Teflon square digestion vessels at 160 °C for 2 days. Following the evaporation of HNO3 and HF, the samples were subsequently treated with HNO3 and HCl (1:3) and refluxed at 80 °C for 1 day. Then the samples were dried down and re-dissolved in 1 mL concentrated HCl at 130 °C. Finally, the samples were dried down again and brought into solution in 8 M HCl (+0.001% H2O2) for chemical purification. Fe and Zn were purified using a two-step ion exchange chromatography. The solution was loaded onto pre-conditioned 2 mL Bio-Rad AG-MP-1M resin first. Matrix elements were eluted in the next 9 mL of 8 M HCl. Fe fraction was collected in 18 mL of 2 M HCl. After that, 2 mL of 0.5 M HNO3 was added to resin and Zn fraction was collected in 10 mL of 0.5 M HNO3 sequentially. Fe and Zn fractions were evaporated to dryness and then redissolved in 1 mL of 8 M HCl (+0.001% H2O2) separately for second chemical purification. They were loaded onto pre-conditioned 1 mL Bio-Rad AG-1-X8 resin for second purification. 9 mL of 8 M HCl was used to elute the matrix elements and Fe fraction was collected in the next 10 mL of 0.4 M HCl. Zn fraction was collected in 7 mL of 0.5 M HNO3 afterwards. The final Fe and Zn eluates were evaporated to dryness and diluted in 2% HNO3 to 30 ng g-1 (Fe) or 500 ng g-1 (Zn) for isotopic measurements. The total procedure blank for Fe and Zn isotope analyses are 5 ng and 6 ng, respectively.
Fe and Zn isotopic measurements were performed on Nu Sapphire CC-MC-ICP-MS at the conventional pathway (Zn) or the collision cell pathway (Fe) in low resolution mode using the sample-standard bracketing method. Data were collected in static mode, with connected to pre-amplifiers fitted with 1011 Ω resistor. Each analysis consisted of a block with 50 cycles of 3 s integration. A 30 s (Zn) or 50 s (Fe) wash was performed in 2% HNO3 between each standard and sample to avoid cross contamination. Long-term reproducibility is 0.03‰ (2sd) for Fe and Zn isotopic measurements. The Fe and Zn isotope results were reported as the per mil deviation relative to standard IRMM-014 and JMC 3-0749L, respectively. The results of samples are presented in table 1, and geostandards in table 2. The values of geostandards are consistent with previous results (J. Wang et al., 2022, 2023 and references therein).
4. Results
The Fe isotope compositions of the Purang ophiolite suite reveal mineral-scale fractionations across lherzolites, harzburgites, dunites and chromitites (fig. 3; table 1). In lherzolites, olivine is enriched in heavy Fe isotopes, with δ56Fe values ranging from 0.05‰ to 0.08‰, while coexisting spinel is isotopically lighter, with δ56Fe values between –0.10‰ and –0.06‰. Orthopyroxene exhibits intermediate δ56Fe values from –0.15‰ to 0.00‰, and clinopyroxene shows much higher δ56Fe values of 0.04‰ to 0.17‰. Harzburgites display a similar pattern: olivine δ56Fe ranges from 0.02‰ to 0.14‰, spinel from –0.18‰ to –0.04‰, orthopyroxene from –0.13‰ to 0.01‰, and clinopyroxene from 0.06‰ to 0.15‰. In dunites, olivine δ56Fe spans –0.08‰ to 0.12‰, and spinel ranges from –0.12‰ to 0.04‰. Chromitites show olivine δ56Fe values from –0.03‰ to 0.15‰ and spinel δ56Fe from –0.02‰ to 0.07‰.
Zn isotope compositions mirror the Fe isotope trends, with a clear light-heavy partitioning (fig. 3; table 1). In lherzolites, olivine δ66Zn spans 0.20‰ to 0.31‰, while spinel is isotopically light, with δ66Zn of –0.25‰ to –0.20‰. Orthopyroxene has moderate δ66Zn values between 0.12‰ and 0.21‰, and clinopyroxene ranges from 0.09 ‰to 0.13‰. In harzburgites, Zn isotopic compositions of minerals vary from 0.11‰ to 0.37‰ for olivine, from –0.27‰ to –0.06‰ for spinel, from 0.05‰ to 0.26‰ for orthopyroxene, and from 0.09‰ to 0.42‰ for clinopyroxene. Dunites exhibit greater scatter in olivine δ66Zn (0.02‰ to 0.29‰), with spinel ranging from –0.21‰ to –0.01‰. In chromitites, olivine δ66Zn spans 0.09‰ to 0.31‰, while spinel δ66Zn ranges from –0.10‰ to 0.00‰.
5. Discussion
5.1. Widespread reversed inter-mineral fractionation of Fe-Zn isotopes in oceanic lithospheric mantle
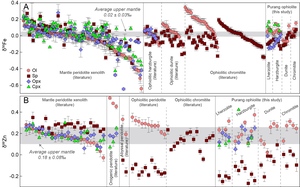
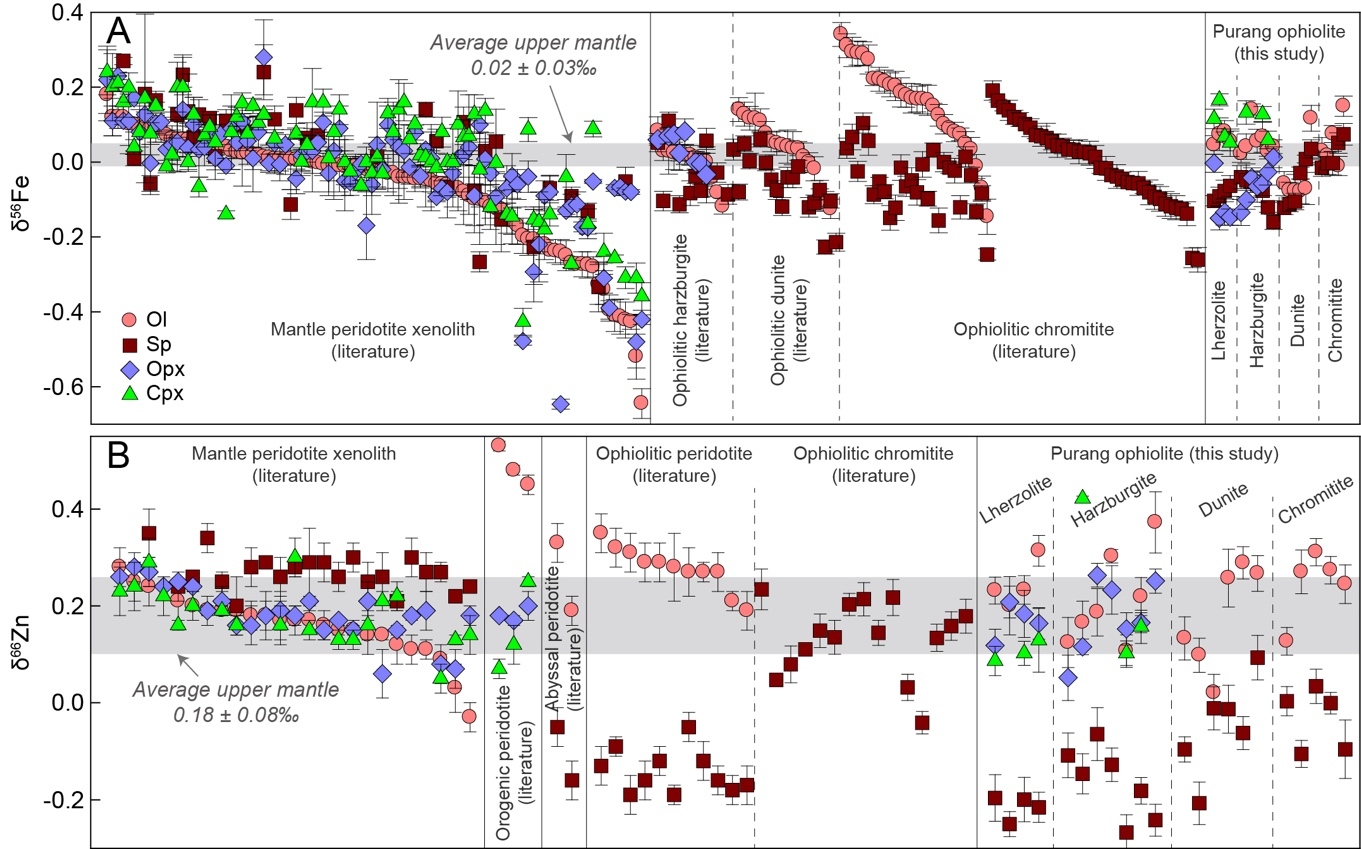
Global isotope data compilations highlight a consistent pattern of reversed Fe-Zn isotope fractionation in oceanic lithospheric mantle, distinct from continental mantle peridotite xenoliths (fig. 3). In ophiolitic lherzolites, harzburgites, dunites, and chromitites, olivine exhibits δ56Fe values (fig. 3A) typically higher than or within the average upper mantle range (0.02 ± 0.03‰; Weyer & Ionov, 2007), while coexisting spinel displays a broader δ56Fe range, predominately lighter than this benchmark (fig. 3A). The Purang ophiolite reinforces this trend, with olivine isotopically heavier than spinel, and clinopyroxene showing notably elevated δ56Fe values compared to other coexisting minerals (table 1). Orthopyroxene δ56Fe values generally exceed those of spinel, though some outliers with lower δ56Fe are noted and discussed later. This silicate > spinel δ56Fe sequence in ophiolitic rocks inverts the bonding-strength-driven equilibrium fractionation (Macris et al., 2015; Roskosz et al., 2015; Schauble et al., 2001) observed in mantle peridotite xenoliths (fig. 3A).
Zinc isotopes exhibit even sharper contrasts. In mantle peridotite xenoliths, spinel is enriched in heavier Zn isotopes relative to olivine, orthopyroxene and clinopyroxene, all of which align closely with the average upper mantle δ66Zn (0.18 ± 0.08‰; Sun et al., in press) (fig. 3B) and follow bonding-strength predictions (Fang et al., 2022; Z.-Z. Wang et al., 2017; Yang et al., 2021). Conversely, ophiolitic spinel and olivine show δ66Zn values extending to significantly lighter and heavier compositions, respectively, far beyond the average upper mantle range (fig. 3B). While pyroxene δ66Zn values in ophiolitic rocks resemble those in xenoliths (Fang et al., 2022; Z.-Z. Wang et al., 2017), their consistently heavier compositions than coexisting spinel distinguish oceanic from continental mantle mineral pairs (fig. 3B).
This reversed Fe and Zn isotope fractionation is not exclusive to ophiolites but also evident in abyssal and orogenic peridotites, albeit with limited data (Fang et al., 2022; Yang et al., 2021). Abyssal peridotites mirror ophiolitic olivine-spinel Zn isotope patterns, while orogenic peridotites show heavy olivine and comparable pyroxene δ66Zn relative to ophiolitic counterparts (fig. 3B). Given that ophiolitic, abyssal, and orogenic peridotites are all representative of oceanic lithospheric mantle, this reversed inter-mineral fractionation appears widespread, pointing to a process distinct from equilibrium fractionation or simple melt/fluid-rock interaction. Notably, the magnitude of inter-mineral isotope fractionation in oceanic lithospheric mantle surpasses that in continental settings (fig. 3), implying a potentially fundamental geochemical distinction between them.
5.2. Effects of grain size and serpentinization on mineral Fe-Zn isotopes
In the Purang ophiolite, most samples exhibit reversed Fe-Zn isotope fractionation between silicate and spinel, but certain lherzolites and harzburgites (e.g., samples PL14-5-7 and PL14-5-6) display orthopyroxene with lower δ56Fe than coexisting spinel (fig. 4A). Petrographic analysis reveals that these samples contain unusually large orthopyroxene grains (0.5–2.5 cm) compared to others (< 0.5 cm) (figs. 2B, C, S1). The large grain size likely limited isotope modification, preserving a δ56Fe closer to primary compositions. In contrast, Zn isotope compositions of these orthopyroxene grains align with those of smaller grains (fig. 3B), likely due to Zn’s lower abundance relative to Fe, which facilitates greater isotope sensitivity to modification.
Serpentinization also influences isotope signatures in some Purang samples. Serpentinized dunites and chromitites (e.g., PL14-5-19, PL14-5-8; figs. 2F, H, S1) show lighter Fe isotope compositions in olivine compared to coexisting spinel (fig. 4A). This shift likely results from serpentinization-driven oxidation and preferential leaching of heavier Fe isotopes from olivine (Scott et al., 2017; B.-X. Su et al., 2015). Zinc isotopes, unaffected by valence changes, exhibit minimal fractionation during serpentinization (fig. 3B; S.-A. Liu et al., 2019; Yang et al., 2021). Excluding these outliers, Purang minerals display a positive correlation between δ56Fe and δ66Zn (fig. 4B), reflecting similar geochemical behaviors of Fe and Zn isotope systematics.
Mineral chemistry further informs isotope variability. Spinel, rich in total FeO (fig. S2A), Fe3+, and Zn (500–1700 ppm) compared to olivine (intermediate FeO, Zn ~20–95 ppm) and pyroxenes (Zn ~10–30 ppm; B. X. Su et al., 2019), is less abundant but more resistant to isotope modification. Olivine, dominant in modal abundance, lacks Fe3+ and shows larger Fe-Zn isotope variations (figs. 3, 4B).
5.3. Influences of partial melting, melt metasomatism, and magma differentiation on inter-sample Fe-Zn isotope variations
5.3.1. Partial melting and melt metasomatism in peridotites
Both continental and oceanic lithospheric mantle undergo partial melting and metasomatism, which influence Fe and Zn isotope compositions. Partial melting typically enriches residual rocks and minerals in lighter isotopes (Z.-Z. Wang et al., 2017; Weyer & Ionov, 2007), while melt metasomatism normally introduces heavier compositions to re-equilibrated phases (Sun et al., in press; Weyer & Ionov, 2007). These processes primarily contribute to inter-sample isotope variability. Spinel Cr# and olivine FeO content serve as proxies for mantle melting and metasomatism. Spinel from mantle peridotite xenoliths and ophiolitic peridotites, including Purang samples, shows a negative correlation between δ56Fe and Cr#, suggesting retention of partial melting and metasomatism signatures (fig. 4C). The scattered distribution of xenolithic spinel likely reflects metasomatic melts with variable Fe isotope compositions (Poitrasson et al., 2013; Zhao et al., 2012, 2015). In contrast, olivine Fe isotope compositions show no clear correlations with FeO content in either xenoliths or ophiolites (fig. S2). Limited Zn isotope data for xenolithic olivine align with partial melting- metasomatism trends, whereas olivine from ophiolitic, abyssal and orogenic peridotites deviates toward lower FeO and higher δ66Zn values (fig. 4D). This indicates that peridotitic olivine in oceanic lithospheric mantle is more susceptible to isotope disturbance, preserving fewer primary melting or metasomatism signatures compared to spinel (Pan et al., 2025; B.-X. Su et al., 2018, 2020, 2023; Zhang et al., 2019, 2024). However, neither processes can reverse the inter-mineral isotope sequence as observed in ophiolitic rocks (fig. 3).
5.3.2. Effect of magma differentiation on mineral isotopes of chromitites and dunites
Chromitites and dunites in ophiolites, being of cumulate origin in magma chambers and conduits (e.g., B.-X. Su et al., 2020, 2023, 2026), exhibit distinct isotope trends. Spinel in these rocks shows increasing δ56Fe and δ66Zn with higher Fe3+/(Fe3++Fe2+) ratios, a marker of magma differentiation (fig. 4E). These spinels are distinguished from those in peridotite xenoliths and oceanic mantle peridotites by intermediate δ66Zn values and FeO contents (fig. S2C). Cumulus olivine Zn isotopes follow magma differentiation trends (fig. 4D), but their δ56Fe values diverge from this path (fig. 4F), suggesting that significant post-crystallization isotope modification in olivine, which is discussed below.
5.4. Influences of subsolidus exchange and hydrous fluid-mediated diffusion on reversed inter-mineral Fe-Zn isotopes
Element diffusion, particularly during subsolidus exchange, is a well-established mechanism for isotope fractionation, with lighter isotopes diffusing faster than heavier ones (e.g., Dauphas et al., 2010; Sio et al., 2013; Teng et al., 2011). This process accounts for the observed Fe, Zn, Cr, and Mg isotope fractionation between spinel and olivine in layered intrusions (Bai et al., 2019, 2021) and ophiolites (Xiao et al., 2016, 2023). During subsolidus exchange, Fe and Cr migrate from olivine to spinel, while Mg moves in the opposite direction, resulting in elevated δ56Fe and δ53Cr values and reduced δ26Mg in olivine, with converse trends in spinel. The extent of fractionation depends on mineral modal abundances, spatial relationships, and element concentrations (Pagé & Barnes, 2009; B.-X. Su et al., 2021, 2026; Xiao et al., 2016, 2023). In peridotites, low abundances of spinel (<5%) and their high FeO and Zn contents make it highly susceptible to isotope modification, while in chromitites, variable abundances of olivine and their lower FeO and Zn contents amplify its sensitivity to exchange (figs. 2E–H, S2). Olivine in oceanic lithospheric mantle peridotites and dunite cumulates deviates significantly from partial melting and metasomatism trends, aligning instead with patterns of element exchange (figs. 4D, F, S2B). Spinel in ophiolitic chromitites shows notably δ56Fe variability at a given Fe3+/(Fe3++Fe2+) ratio, consistent with subsolidus exchange effects (fig. 4E).
High-temperature dry diffusion (e.g., subsolidus Fe-Mg exchange) can produce reversed fractionation (Bai et al., 2019, 2021; Xiao et al., 2016) but is limited by grain boundary distances (<1 mm) and requires long residence times (>107 years at 900 °C; Sio et al., 2013), which fails to account for the widespread reversal in ophiolites lacking close mineral contacts (fig. 5A–C). Deformation-enhanced diffusion (e.g., via dislocations) may amplify rates but still relies on proximity and does not explain pervasive patterns across unstrained samples (e.g., Purang lherzolites; fig. 2). Oxygen fugacity changes (e.g., fO2 increase during serpentinization) affect Fe valence and isotopes but not Zn, which lacks redox sensitivity (S.-A. Liu et al., 2019; Muir et al., 2023). The coupled reversed Fe-Zn (and Cr) fractionation uniquely points to hydrous fluid mediation, as fluids enable long-range transport and selective element mobility (e.g., Fe2+ and Zn substituting Mg in lattices; Lian et al., 2024; Yang et al., 2021).
We note that H2O can significantly enhance diffusion rates in nominally anhydrous minerals at subsolidus temperatures (800–1000 °C), as demonstrated by experimental studies (e.g., Demouchy & Bolfan-Casanova, 2016; Nishi, 2015). Hydrous fluids facilitate element mobility over distances of centimeters to meters, far exceeding dry subsolidus diffusion limits (e.g., Bai et al., 2021; Sio et al., 2013). Experimental diffusion models indicate that H2O contents of 10–50 ppm increase Fe-Mg interdiffusion coefficients in olivine by 1–2 orders of magnitude (e.g., Hier-Majumder et al., 2005), sufficient to produce the observed Δ56Fesilicate-spinel of 0.1–0.3‰ over timescales of 104–106 years at 900 °C. Consequently, to account for the pervasive reversed fractionation, hydrous fluid-assisted diffusion is critical (B. X. Su et al., 2021; B.-X. Su et al., 2021). Highly mobile hydrous fluids overcome spatial barriers of subsolidus exchange, facilitating large-scale element transport across minerals (fig. 5A–C). This process drives significant Fe enrichment in spinel and depletion in olivine, particularly pronounced in peridotites and chromitites, respectively (fig. 5A–C; B. X. Su et al., 2021). Zinc, with geochemical behavior similar to Fe, is co-mobilized or substitute for Mg in mineral lattices, leading to lighter Fe-Zn isotope compositions in spinel relative to silicates (fig. 4; Lian et al., 2024; Yang et al., 2021). Petrographic evidence, including spinel zoning and inter-grain compositional heterogeneity, supports hydrous fluid-mediated exchange (Gamal El Dien et al., 2019; Pan et al., 2022; B.-X. Su et al., 2023, 2026). Elevated H2O contents in ophiolitic olivine, positively correlated with Fo values, further confirm hydration-driven compositional changes (Cui et al., 2025; B.-X. Su et al., 2025, 2026). Additionally, lower δ18O values in ophiolitic spinel (1.04–4.87‰) compared to silicates (e.g., olivine: 4.90–6.68‰) in dunites and harzburgites indicate extensive fluid interaction (Pan et al., 2025). The intergrowth of clinopyroxene and spinel in ophiolitic rocks (B. X. Su et al., 2019) results in comprehensive element exchange and thus heavier Fe-Zn isotope compositions in clinopyroxene (fig. 3). These observations suggest that spinel in ophiolitic mantle peridotites rarely retains pristine paleo-oceanic mantle signatures, highlighting its sensitivity to fluid-mediated modification and challenging its use as a reliable petrogenetic indicator. We acknowledge that these estimates are based on representative ophiolitic data, future direct analyses of H2O contents on our mineral separates would provide even tighter constraints on sample-specific variability.
5.5. Extensive hydration in oceanic lithospheric mantle
Recent studies of ophiolitic peridotites have revealed hydration as a pervasive and transformative process in the oceanic lithospheric mantle, with profound implications for its geochemical and physical properties. Traditionally viewed as a minor, surface-related phenomenon associated with hydrothermal alteration or late-stage serpentinization (Greenberger et al., 2021; Gruau et al., 1998; Sanfilippo et al., 2025), hydration is now recognized as a deep-seated process (Cui et al., 2025; Demouchy et al., 2015; Nishi, 2015; Schmädicke et al., 2018; B.-X. Su et al., 2023, 2025). Isotopic evidence, including reversed Fe and Zn isotope fractionation between spinel and silicates, contrasting with equilibrium patterns in continental mantle xenoliths, indicates fluid-mediated element exchange as a common feature of oceanic mantle rocks (ophiolites, abyssal, and orogenic peridotites) (fig. 3; Chen et al., 2019; Fang et al., 2022; Yang et al., 2021). Petrographic features, such as zoning in spinel, compositional heterogeneity, and variable oxygen isotope ratios (e.g., spinel δ18O: 1.04–4.87‰ vs. olivine: 4.90–6.68‰), alongside the presence of hydrous minerals like amphibole, confirm widespread hydrous fluid infiltration (Gamal El Dien et al., 2019; C.-Z. Liu et al., 2010; Pan et al., 2022, 2025; B.-X. Su et al., 2020, 2023, 2026).
Hydrous fluids in the oceanic mantle originate from multiple sources. In supra-subduction zone (SSZ)-type ophiolites like Purang, slab-derived fluids dominate during forearc extension (post-melting, pre-obduction) (C.-Z. Liu et al., 2010; Pan et al., 2025; B.-X. Su et al., 2023). In contrast, mid-ocean ridge (MOR)-type abyssal peridotites may involve asthenospheric fluids at the lithosphere base (Hirschmann & Kohlstedt, 2012). Post-obduction serpentinization cannot explain the coupled Fe-Zn reversal, as it primarily affects Fe isotopes via oxidation without reversing Zn isotope fractionation (S.-A. Liu et al., 2019). Chromitite formation releases hydrous fluids, as observed in ophiolites (Cui et al., 2025; Matveev & Ballhaus, 2002; B. X. Su et al., 2021; B.-X. Su et al., 2020, 2021, 2025, 2026) and layered intrusions like the Stillwater Complex, which exhibits reversed Fe-Cr isotope fractionation (Bai et al., 2019, 2021, 2024). In ophiolites lacking significant chromite deposits, such as Purang, subduction-related fluids from slab dehydration infiltrate the mantle wedge, driving hydration and isotopic resetting (Gose & Schmädicke, 2021; B.-X. Su et al., 2023). Additionally, asthenospheric upwelling introduces minor hydrous fluids due to water gradient (Hirschmann & Kohlstedt, 2012), further hydrating the lithosphere base (Cui et al., 2025; B.-X. Su et al., 2025). These diverse sources underscore the complex, multi-stage nature of mantle hydration.
6. Conclusion
While pronounced in SSZ ophiolites like Purang, the reversal of inter-mineral Fe-Zn isotope fractionation is evident in MOR-type abyssal peridotites (albeit with fewer data; Yang et al., 2021), indicating a potentially fundamental oceanic lithospheric mantle trait. Future studies on pristine abyssal samples will test its ubiquity. Driven by diverse fluid sources, including chromitite formation, subduction-related dehydration, and asthenospheric upwelling, this hydration facilitates extensive fluid-mediated element exchange, profoundly altering mantle geochemistry. Historically underestimated due to methodological limitations and an overemphasis on anhydrous processes, hydration is now recognized as a deep-seated, integral component of mantle evolution, impacting supra-subduction and likely abyssal settings. Its effects extend beyond geochemical overprinting to influence mantle rheology, volatile cycling, and geophysical properties. Recognizing the scale and significance of hydration is critical for refining models of oceanic lithospheric mantle formation, subduction zone dynamics, and global tectonic and geochemical evolution. Hydration emerges not as a secondary process but as a primary architect of the oceanic mantle’s physiochemical framework.
Acknowledgments
We appreciate two anonymous reviewers for their constructive comments, which significantly improve the quality of the paper. This study was supported by special fund of Deep Earth Probe and Mineral Resources Exploration-National Science and Technology Major Project of China (2025ZD1006104), National Natural Science Foundation of China (92462305, 42350001), and China Geological Survey ((2025)02-032-01-06).
Author contributions
B.-X.S. and Y.X. designed and led the project. J.W., S.-Q.Z. and Q.-Q.P. conducted the analyses. B.-X.S. wrote the manuscript with inputs from Y.X., J.W., Q.-Q.P., and S.-Q.Z. All authors contributed the interpretation of the results.
Data availability
Data are available through https://figshare.com/ at http://10.6084/m9.figshare.29436158.
Supplemantary information
https://doi.org/10.17632/y6sz2n5tys.1
Editor: C. Page Chamberlain, Associate Editor: Guochun Zhao

_tectonic_sketch_map_of_tibetan_plateau__highlighting_major_suture_zones._(b)_distribut.jpeg)
_fe_isotope_compositions_of_minerals_in_samples_from_the_purang_ophiolite__with_sample_.jpeg)
_schematic_model_of_fluid-assisted_element_exchange_and_subsolidus_diffusion_in_vari.jpeg)
_tectonic_sketch_map_of_tibetan_plateau__highlighting_major_suture_zones._(b)_distribut.jpeg)
_fe_isotope_compositions_of_minerals_in_samples_from_the_purang_ophiolite__with_sample_.jpeg)
_schematic_model_of_fluid-assisted_element_exchange_and_subsolidus_diffusion_in_vari.jpeg)